 
When on the institution site, please use the credentials provided by your institution.Select your institution from the list provided, which will take you to your institution's website to sign in.Click Sign in through your institution.Shibboleth/Open Athens technology is used to provide single sign-on between your institution’s website and Oxford Academic. 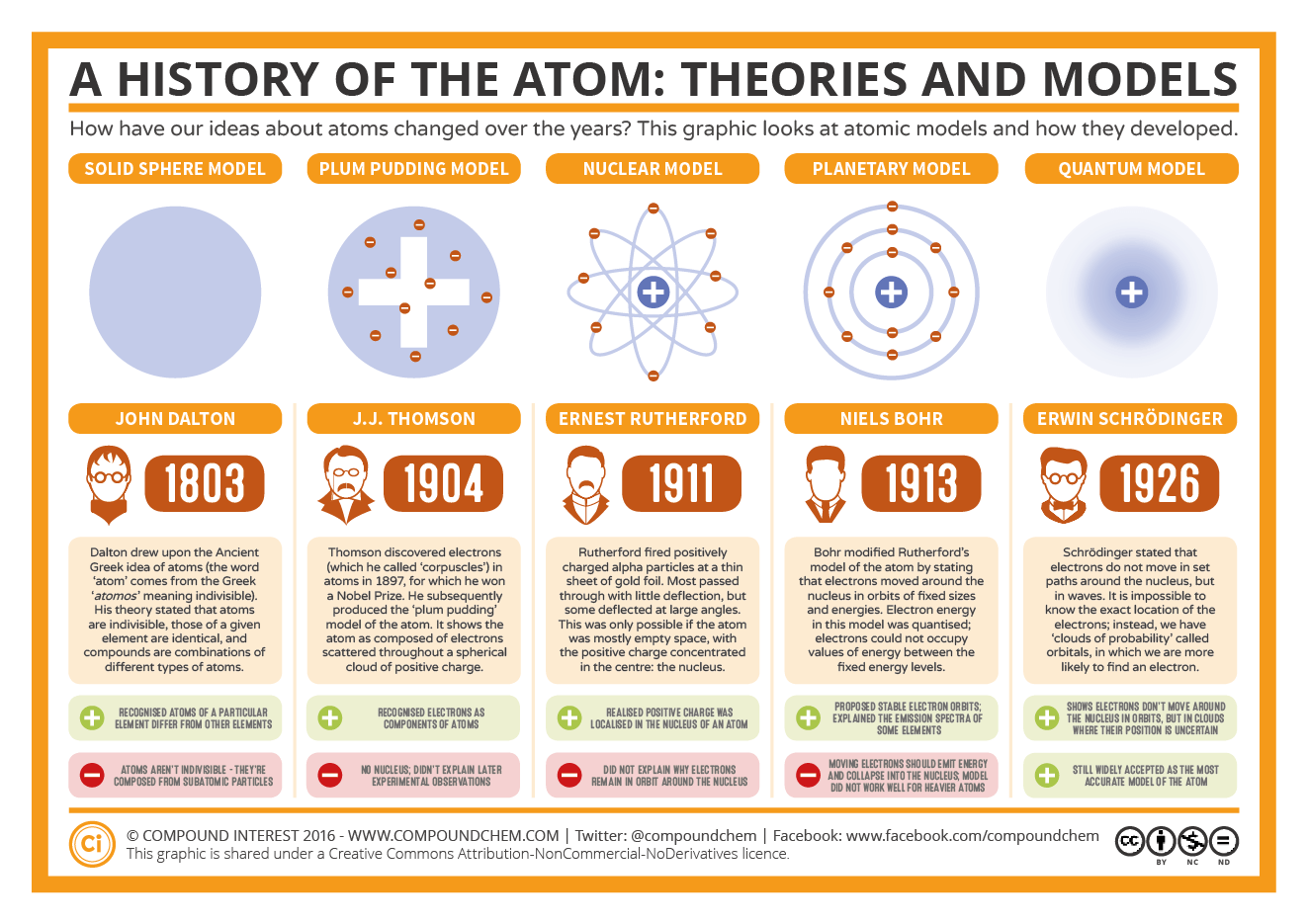
This authentication occurs automatically, and it is not possible to sign out of an IP authenticated account.Ĭhoose this option to get remote access when outside your institution. Typically, access is provided across an institutional network to a range of IP addresses. If you are a member of an institution with an active account, you may be able to access content in one of the following ways: Get help with access Institutional accessĪccess to content on Oxford Academic is often provided through institutional subscriptions and purchases. By discussing the theory in its entirety-following it from its birth in 1913, through its adolescence round 1918, to its decline in 1924-it becomes possible to understand its development and use it as an example of the dynamics of scientific theories. The book covers the successes as well as the failures of Bohr’s theory, arguing that the latter were no less important in the process that led Bohr to abandon the original model and Heisenberg to propose a new ‘quantum mechanics’. It, namely, as an ambitious attempt to understand the structure of atoms as well as molecules: the chemical aspects of the theory are given much attention. Moreover, it discusses the theory as Bohr originally conceived Contrary to most other accounts of the Bohr atom, the book presents it in a broader perspective, which includes the reception among other scientists, popular expositions of the theory, and the objections raised against it by scientists of a more conservative inclination. By following the development and applications of the theory, it brings new insight into Bohr’s peculiar way of thinking what Einstein once called his ‘musicality’ and ‘unique instinct and tact’. It offers a comprehensive account of Bohr’s ideas and the way they were modified by other physicists. This is the first book that focuses in detail on the origin and development of this remarkable theory. S2CID 250900220.The theory of atomic structure proposed by the young Danish physicist Niels Bohr in 1913 marked the true beginning of modern atomic and quantum physics. "Sommerfeld formula and Dirac's theory" (PDF). ^ - Atombau und Spektrallinien, 1921, page 520."The Kossel-Sommerfeld Theory and the Ring Atom". "Einstein's unknown insight and the problem of quantizing chaos" (PDF). ^ The Collected Papers of Albert Einstein, vol."The quantum theory of radiation and line spectra". Nevertheless, both solutions fail to predict the Lamb shifts. This solution (using substitutions for quantum numbers) is equivalent to the solution of the Dirac equation. The Bohr–Sommerfeld model supplemented the quantized angular momentum condition of the Bohr model with an additional radial quantization condition, the Wilson– Sommerfeld quantization condition ∫ 0 T p r d q r = n h, is the fine-structure constant. Sommerfeld argued that if electronic orbits could be elliptical instead of circular, the energy of the electron would be the same, except in the presence of a magnetic field, introducing what is now known as quantum degeneracy. Bohr–Sommerfeld theory is named after Danish physicist Niels Bohr and German physicist Arnold Sommerfeld. The Bohr–Sommerfeld model (also known as the Sommerfeld model or Bohr–Sommerfeld theory) was an extension of the Bohr model to allow elliptical orbits of electrons around an atomic nucleus. The Sommerfeld extensions of the 1913 solar system Bohr model of the hydrogen atom showing the addition of elliptical orbits to explain spectral fine structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
